 
For example, a quick glance at Figure AT5.2. However, as you go down the periodic table, there are more filled shells, so the valence electron are further away from the nucleus, and so are not as tightly held in, increasing the atomic radius. That way, we can get a better look at the relationship. In the same group, the effective nuclear charge does not change. As we will see below, the periodic table organizes elements in a way that reflects their number and pattern of electrons, which makes it useful for predicting the reactivity of an element: how likely it is to form bonds, and with which other elements. Often it is useful to plot data on a graph. As a result, fluorine is usually thought of as the most electronegative element. However, on many scales, fluorine would be the most electronegative atom here. As a result, noble gases are also given electronegativity values on this scale. 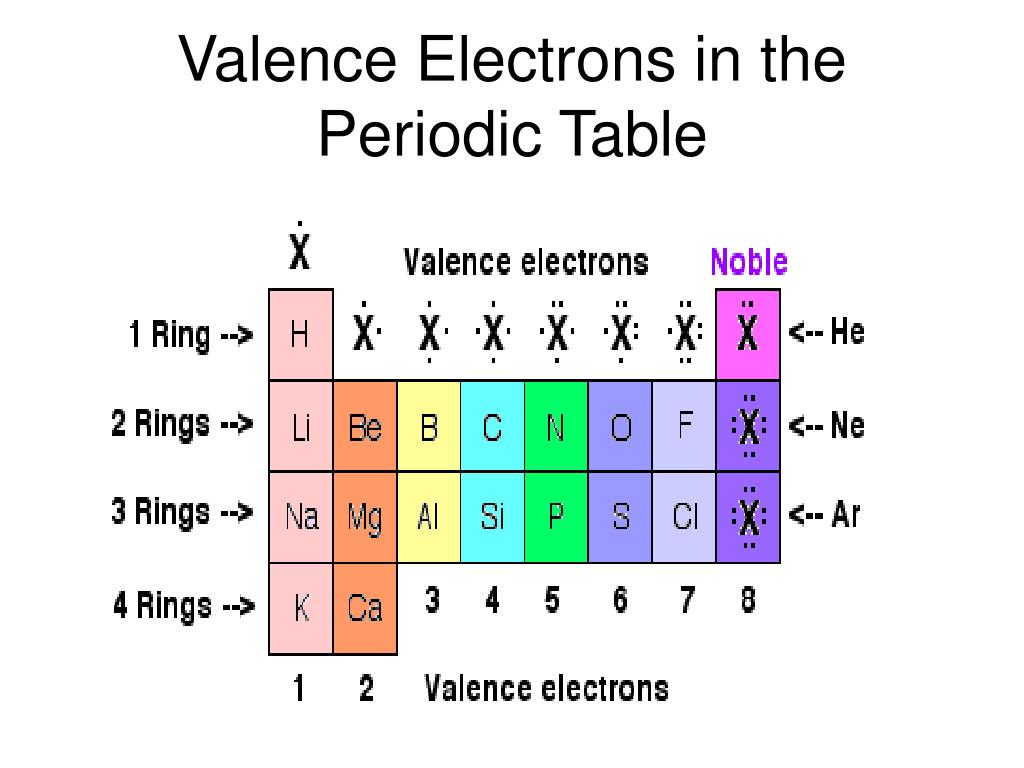 
The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. \): The Allen electronegativity values of the second-row elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
